HIV: How a Microscopic Virus Outsmarts Human Immunity
The Human Immunodeficiency Virus (HIV) continues to be one of the most significant viral pathogen changing public health worldwide, affecting about 40.4 million people, including 1.5 million children. Since the epidemic began, approximately 40.1 million people have died from HIV-related causes. HIV-1 is the most dangerous and widespread variety globally, whereas HIV-2 is most prevalent in West Africa (Swinkels et al., 2025). Without treatment, HIV advances to Acquired Immunodeficiency Syndrome (AIDS), a fatal condition characterized by profound immune suppression. There are 11 phases of the HIV life cycle. HIV is an enveloped virus containing two single-stranded RNA genomes and essential enzymes like reverse transcriptase and integrase. It cannot survive long outside the human body, as it is highly sensitive to temperature, desiccation, and disinfectants (Sarafianos et al., 2009). Its environmental fragility underscores the importance of direct contact for transmission. This blog article explores HIV transmission mechanisms and its complex relationship with the host immune system, which are crucial for developing effective prevention and treatment strategies.
HIV Transmission Mechanisms
HIV is transmitted through direct contact with infected bodily fluids that contain a sufficient viral load. The main modes of transmission are sexual contact, parenteral exposure, and vertical transmission from mother to child (Huynh et al., 2025). Sexual transmission is the most common, accounting for about 80% of new infections globally. It occurs through vaginal, anal, or oral sex, especially when infected fluids contact mucous membranes or open wounds. Anal intercourse poses the highest risk due to the fragility of rectal tissues. Factors such as high viral load, presence of other STIs, and lack of male circumcision can increase susceptibility (Broyles et al., 2023). Parenteral transmission, particularly through contaminated needles or unscreened blood products, remains a major concern, especially among intravenous drug users (Scott and Wu, 2019). Mother-to-child transmission can happen during pregnancy, childbirth, or breastfeeding. However, with effective antiretroviral therapy, the transmission risk can be reduced from 15-45% to below 2% (Dong et al., 2020).
Host Immune Response to HIV
The human immune response to HIV involves a complex interplay between the innate and adaptive immune systems. Despite their coordinated actions, HIV has evolved mechanisms to persist and eventually overwhelm these defenses.
When HIV enters the body, innate immune system cells such as dendritic cells, macrophages, and natural killer cells (NK cells) become activated. Pattern recognition receptors (PRRs), which include Toll-like receptors (TLRs) and RIG-I-like receptors (RIG-I and MDA5) recognize HIV RNA. This triggers signaling pathways involving MAVS, TRAF3, and IRF3, which stimulate the production of type I interferon (IFN-α and IFN-β). These interferon connect to cell surface receptors and regulate the JAK-STAT pathway, causing the production of interferon-stimulated genes (ISGs) that impede viral replication (Masenga et al., 2023). Dendritic cells are critical for connecting both the adaptive and innate immune responses. Plasmacytoid DCs produce high levels of IFN-α via TLR7 detection of HIV RNA, while conventional DCs secrete cytokines like IL-6 and IL-12 and present HIV antigens to T cells. Macrophages contribute by engulfing viral particles and releasing inflammatory cytokines (Kwaa and Blankson, 2024). They also activate autophagy and inflammasome pathways to limit infection.
In adaptive immunity, CD4+ T cells despite being the main targets of HIV activate B cells and support CD8+ T cell responses. Over time, their depletion weakens immune coordination. CD8+ T cells kill infected cells via MHC class I recognition and release of perforin and granzymes, along with antiviral cytokines such as IFN-γ (Masenga et al., 2023). B cells eventually generate HIV-specific antibodies, including neutralising types. However, these are delayed and often compromised due to HIV’s rapid mutation and immune evasion strategies, limiting their long-term effectiveness (Figure 1).
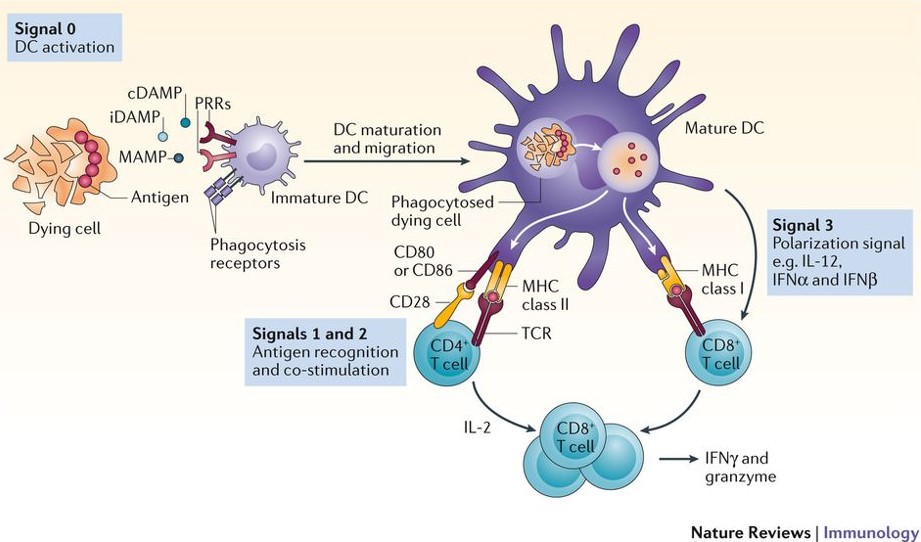
Figure 1-Innate immune responses and the initiation of adaptive immunity (“HIV Immunology,” 2019)
Screening, Diagnosis and Treatment of HIV
HIV diagnosis relies on detecting viral components and the host's immune response. Fiebig staging classifies early infection into six phases based on viral and antibody markers. Seroconversion, or the appearance of HIV-specific antibodies, usually occurs within 4–6 weeks post-infection. ELISA tests detect antibodies and viral proteins, while Western blotting confirms diagnosis. From Fiebig stage III, HIV-specific antibodies become detectable (Saag, 2021). Immunological evaluation comprises assessing blood lymphocyte fragments (CD3+, CD4+, and CD8+) and the CD4/CD8 ratio. A CD4+ T cell count < 200 cells/mm³ indicates AIDS, typically reached within 10 years without treatment, indicating severe immunosuppression (Fred, 2018).
Although HIV cannot be eliminated, antiretroviral medication (ART), particularly Highly Active Antiretroviral Therapy (HAART), effectively lowers viral replication and reduces the progression of the infection. HAART includes medications that target various stages of the HIV life cycle. Regular monitoring of CD4 counts and viral load assesses treatment success (Weichseldorfer et al., 2021). Additionally, managing opportunistic infections is crucial, as these are often the primary cause of death in advanced HIV/AIDS.
In conclusion, HIV remains a global health challenge, but effective treatment and prevention strategies have transformed it into a manageable condition. Continued research is vital to achieve a long-term cure and eventual eradication.

